Applications - medical and industrial
The right solution for your application.
.
Here you will find practical application examples. Pantec laser modules can be used for a variety of applications. Due to their compact design and the adjustable performance parameters such as wavelength or pulse rate, our components can be optimally integrated into material processing applications or medical devices. Pantec laser modules are characterized by their quality and robustness.
Would you like to know how our laser modules fit into your application? Contact us! We will gladly answer your questions.

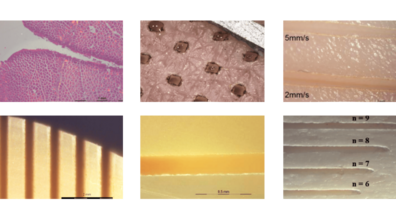
By adjusting parameters such as wavelength or pulse rate, different results and properties are achieved. In this way, laser modules can be adapted to the customer's application to achieve the desired results. 3 m.i.k.r.o.n.TM modules are particularly characterized by their performance, robustness and compact design.
Precise treatments (cutting width <100 µm)
Controllable thermal impact by laser parameters
Cold ablation
(cut like a scalpel)
Hot ablation (coagulation)
by stacked pulses
Limited lateral thermal damage
No unwanted vibrations
Short operation time
Fast healing
Combines benefits of traditional Er:YAG and CO2 lasers
Optimized for soft and hard tissue
Download our application brochures to learn more about the 3 m.i.k.r.o.n.™
technology. The brochures provide information on areas of application and technical data.
Medical applications
Industrial applications
Do you think 3 m.i.k.r.o.n.™ fits to your application?
We are happy to support you in finding the best solution.